|
Abstract
Biological systems often display multiexponential transverse relaxation times, diagnostic of the presence of different proton pools within the sample. A reliable representation of sample multiexponentiality can be calculated through the inversion of CPMG magnetization decays via the UPEN protocol. However, if a sample contains both liquid- and solid-like protons (as for example in muscle tissue or protein gels) CPMG cannot be used, as the solid-like signal may decay before the first echo is recorded. In these cases signal quantification is more simply attempted in the time domain, by fitting the FID to a discrete number of suitable decaying functions, provided no frequency difference exists between the signal pools. Low field spectrometers (0.47 T or less) are useful in this respect because FIDs obtained at low resolution do not usually carry frequency information. On the other side, they may suffer from very poor magnetic field homogeneity - owing to the fact that they are typically built around a permanent magnet with no shimming facilities - thus producing highly distorted FIDs whose shape can be hardly approximated by meaningful fitting functions.
In this work we show that a technique known under the name of reference deconvolution in high resolution NMR can be slightly modified and used successfully in low resolution NMR for fitting highly distorted FIDs. The reference convolution technique starts from the hypothesis that the effect of the magnetic field inhomogeneity on the FID is independent of the sample; an experimental FID (FIDEXP) is thus the point-by-point multiplication of the undistorted FID (FIDUND) by an unknown distortion function D:
(1) 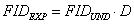 . .
To get rid of D, the FID of a water sample with the same geometry of the previous sample is recorded in the same experimental conditions. The experimental water FID (FIDw) can be expressed as:
(2) 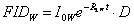 , ,
where R2W is the water transverse relaxation rate. If for example all components of FIDUND are exponential, Equation (1) can be rewritten as:
(3) 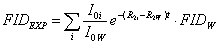 . .
Of course this is equivalent to convolution in frequency domain between an undistorted FID and the water FID acting as a distortion reference, whence the name.
To test the above protocol, the FIDs of two gels obtained by heating at 75 °C two solutions of bovine serum albumine (BSA) in H2O and D2O at pH 8 and equal concentration (respectively A and B in the following figure) were treated according to Eq.(3) using a water reference.
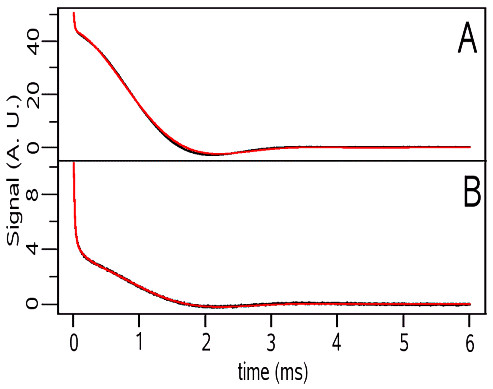
The ratio between the intensity of the fast and the slow components in sample A matched the ratio between the BSA and water protons, thus indicating slow exchange between water and the exchangeable BSA protons, in agreement with previous studies. Further, on passing from H2O to D2O the fast signal decreased by about 17%, an amount comparable to the percentage of exchangeable protons in the BSA. Overall, our results show the usefulness of reference convolution for the removal of field inhomogeneity effects on FID's.
|