|
Permalink of this document: DOI 10.3247/SL5Nmr15.002.
Please, cite this online document as:
Sykora S., Molecular Spins: a New Frontier,
44-th National Congress of GIDRM, Rome (Italy), 28-30 September 2015, DOI: 10.3247/SL5Nmr15.002.
Abstract
The idea presented here falls halfway between 'totally crazy' and 'how comes we have overlooked it'. I have briefly hinted at the idea during the 2011 Italian GIDRM meeting [1], and then in a poster and a talk at the 2012 ENC meeting [2]. This presentation expands on it, adding some more details.
Consider a planar diamagnetic molecule (this whole presentation has nothing to do with molecular paramagnetism) which has an axial sense, such as that of chloraldehyde or, in general C(XYZ), where C is a central atom and XYZ are three different atoms/groups arranged around it (see Fig.1):
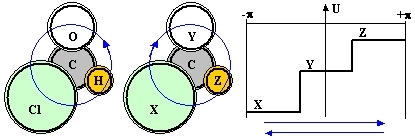
Figure 1: A bonding-shell electron shared by three different atoms XYZ is a bit like a particle constrained to a circular path and moving under the influence of a potential U (solid line) with no circular symmetry. In this case it evidently matters whether its wavefunction propagates clockwise or counter-clockwise (blue arrows) and the energies of the corresponding eigenstates are different.
By itself, the 'axiality' of the arrangement, due to the conceptual difference between a clockwise and a counter-clockwise circumnavigation of the (XYZ) atoms, can not lead to distinct molecular species (such as in chiral arrangements). But it does define an axial vector well oriented with respect to the molecular plane, a fact compatible with the existence of a preferential direction of motion and a persistent electron current loop. A bond electron partially shared by all three atoms can circulate around the structure either in one sense (XYZ), or in the other one (XZY) and, when XYZ are all different, these two types of 'loop orbitals' have distinct energies. Such loop orbitals should mix with the conventional a-circular electron orbitals, convey to them an axial character, and contribute in part to the total energy of the molecule. From the symmetry point of view, the axiality should be accounted for in any math model of the molecule, lest an important feature of the latter is a-priori missed.
The present quantum theory of chemical bonding never considers any axial aspects. They are absent in models based on combinations of electron Slater-type orbitals, because the latter are inherited from two-particle models (electron + nuclide), while axiality requires at least three bodies. Nor are they accounted for in DFT which considers only electron densities, but no persistent electron current loops. In this respect, all present quantum mechanical chemical models could be flawed in their roots, and incapable of accounting for some molecular phenomena.
If the insight is correct then some molecules, just like many elementary particles and many nuclides, should possess an intrinsic spin (an orbital one) and an associated permanent magnetic moment, thus giving rise to magnetic resonance phenomena similar to those we are used to in NMR (in particular, exhibiting in a magnetic field a very specific Larmor frequency). We are therefore talking about the possibility of radically extending the range of 'particles' endowed with spin properties and gyromagnetic moments, and giving birth to Molecular Magnetic Resonance (MMR).
Here I attempt to estimate, starting from first principles, the magnitudes of persistent currents in circular molecules and molecular fragments. Based on the results, I reflect on any molecular spin phenomena that might be observable using the techniques of magnetic resonance and try to delimit the conditions for their detection.
References:
[1] S. Sykora, The Gold Medal Talk at the 40th National Congress of GIDRM, September 26-28, 2011, Parma, Italy.
[2] S. Sykora: Molecular Spins: a new Frontier of Chemical Physics and Magnetic Resonance?,
Poster and Presentation at 53rd ENC, Miami, April 15-20, 2012, DOI: 10.3247/SL4Nmr12.004.
Discussions
Your comments are welcome and will appear here
|